About the Journal
Scope of the Journal
Indexed In
Journal Information
Current issue
Archive
Editorial Board
Journal Policy
APC Policy
Authorship Policy
Author Recognition Policy
Citation Policy
Conflict of Interest (COI) Policy
Copyright Policy
Data Availability Policy
DEI Policy
Digital Archiving Policy
Ethical Approval Policy
Funding Policy
Image Policy
Open Access Policy
Ownership & Governance
Peer Review Policy
Personal Data Protection Policy
Plagiarism Policy
Post-Publication Policy
Preprint Policy
Author Instructions
Submit Manuscript
Contact Information
Journal Policy
APC Policy
Authorship Policy
Author Recognition Policy
Citation Policy
Conflict of Interest (COI) Policy
Copyright Policy
Data Availability Policy
DEI Policy
Digital Archiving Policy
Ethical Approval Policy
Funding Policy
Image Policy
Open Access Policy
Ownership & Governance
Peer Review Policy
Personal Data Protection Policy
Plagiarism Policy
Post-Publication Policy
Preprint Policy
REVIEW PAPER
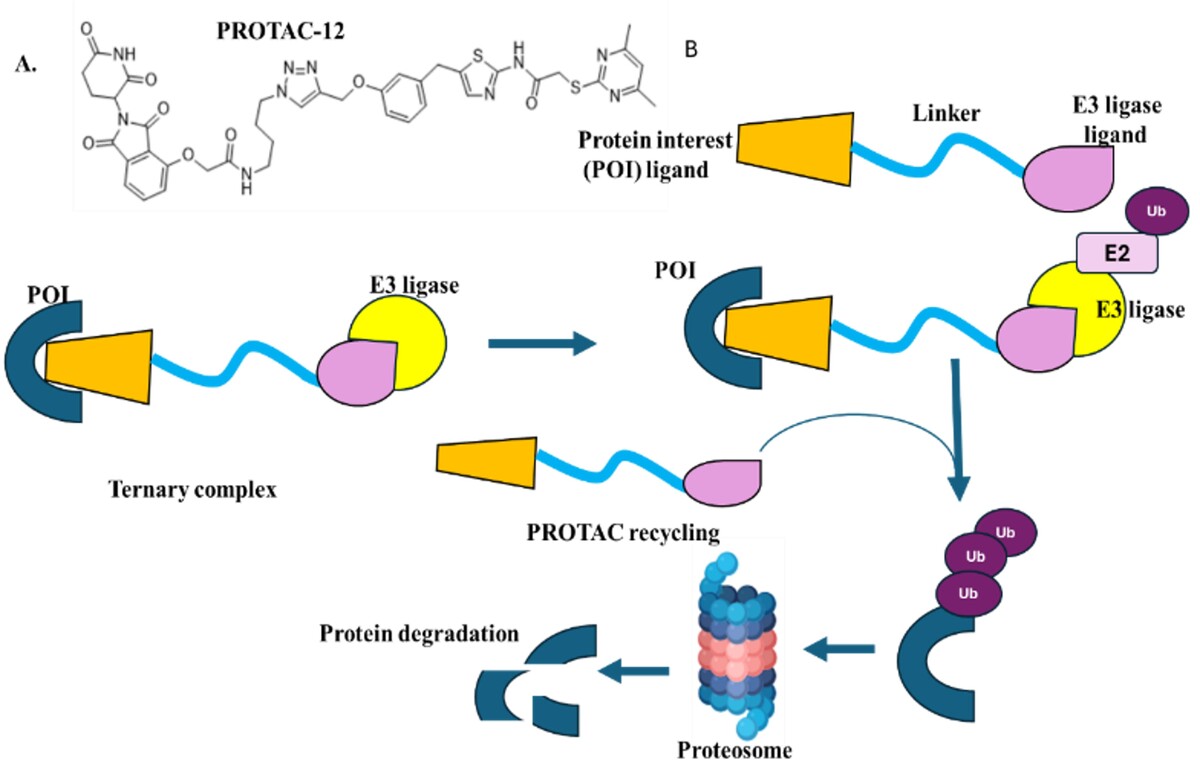
PROTACs in Action: Bridging Chemical Biology and Drug Discovery for Protein Degradation
1
Microbiology, Biochemistry and Immunology, Morehouse School of Medicine, United States
Submission date: 2025-10-23
Final revision date: 2025-12-20
Acceptance date: 2025-12-21
Online publication date: 2026-04-28
Publication date: 2026-04-28
Corresponding author
Ramar Vanajothi
Microbiology, Biochemistry and Immunology, Morehouse School of Medicine, 720, Westview Drive SW, 30310, Atlanta, United States
Microbiology, Biochemistry and Immunology, Morehouse School of Medicine, 720, Westview Drive SW, 30310, Atlanta, United States
Journal of Medico Informatics 2026;02(Issue 02):14-22
HIGHLIGHTS
- PROTACs enable targeted protein degradation via ubiquitin–proteasome system.
- vent-driven pharmacology allows sustained effects with transient drug exposure.
- Heterobifunctional design links target protein and E3 ligase for degradation.
- AI and computational tools accelerate PROTAC design and optimization.
- Clinical applications span oncology, autoimmune, and neurodegenerative diseases.
KEYWORDS
TOPICS
ABSTRACT
Proteolysis-targeting chimeras (PROTACs) represent a transformative paradigm in drug discovery, fundamentally altering how we approach protein-targeted therapeutics by harnessing the ubiquitin-proteasome system for selective protein degradation. This comprehensive review examines the evolution of PROTAC technology from its conceptual origins in 2001 to its current clinical validation, with over 30 molecules in various stages of clinical development as of 2024. We explore the mechanistic foundations of PROTAC action, including the catalytic mode of protein degradation that distinguishes event-driven from occupancy-driven pharmacology, and examine the structural design principles governing heterobifunctional architecture, linker optimization, and E3 ligase recruitment strategies. The review analyzes current therapeutic applications across oncology, autoimmune diseases, and neurodegenerative disorders, highlighting the clinical success of compounds like ARV-471 and ARV-110 in Phase III trials. Critical challenges including physicochemical property optimization, resistance mechanisms, and bioavailability limitations are addressed alongside emerging solutions through computational design, artificial intelligence integration, and next-generation platforms including conditional degraders, nano-PROTACs, and expanded E3 ligase recruitment. Recent advances in molecular glue degraders, which represent 66% of FDA-approved degraders, and the development of precision medicine approaches through biomarker-guided therapy are also examined. The review concludes with an assessment of future directions, including E3 ligase repertoire expansion beyond the current focus on CRBN and VHL, targeting of previously undruggable proteins, and the integration of PROTAC technology with combination therapies and precision medicine strategies.
ABBREVIATIONS
PROTAC – Proteolysis-Targeting Chimera
UPS – Ubiquitin-Proteasome System
E1 – Ubiquitin-Activating Enzyme
E2 – Ubiquitin-Conjugating Enzyme
E3 – Ubiquitin Ligase
POI – Protein of Interest
Ub – Ubiquitin
CRBN – Cereblon
VHL – Von Hippel–Lindau
MDM2 – Mouse Double Minute 2
IAPs – Inhibitors of Apoptosis Proteins
PEG – Polyethylene Glycol
AI – Artificial Intelligence
ML – Machine Learning
MD – Molecular Dynamics
BTK – Bruton’s Tyrosine Kinase
ACKNOWLEDGEMENTS
The author thankfully acknowledge the Microbiology, Biochemistry and Immunology, Morehouse School of Medicine, 720, Westview Drive SW, 30310, Atlanta, United States for providing necessary facilities for performing this study.
FUNDING
This research received no external funding. The study was conducted without any financial support from public, commercial, or not-for-profit funding agencies. All resources utilized for this work were provided by the author respective institutions.
CONFLICT OF INTEREST
The authors declare that there are no conflicts of interest regarding the publication of this article. The authors have no financial, commercial, or personal relationships that could have influenced the work reported in this manuscript.
PEER REVIEW INFORMATION
Article has been screened for originality
© 2026 The Author(s). This article is distributed under the terms of the Creative Commons Attribution License (CC BY 4.0).
REFERENCES (96)
1.
Abeje YE, Wieske LHE, Poongavanam V, Maassen S, Atilaw Y, Cromm P, Lehmann L, Erdelyi M, Meibom D, Kihlberg J. (2025), Impact of Linker Composition on VHL PROTAC Cell Permeability, J Med Chem, 68(1):638-657. doi:10.1021/acs.jmedchem.4c02492. PMID: 39693386.
2.
Agarwal S, McDonald AA, Campbell V, Chen D, Davis J, Rong H, Mishkin A, Slavin A, Gollerkeri A, Gollob JA. (2025), Pharmacokinetics and Pharmacodynamics of KT-474, a Novel Selective Interleukin-1 Receptor-Associated Kinase 4 (IRAK4) Degrader, in Healthy Adults, Clin Transl Sci, 18(3):e70181. doi:10.1111/cts.70181. PMID: 40055981.
3.
An S, Fu L. (2018), Small-molecule PROTACs: An emerging and promising approach for the development of targeted therapy drugs, EBioMedicine, 36:553-562. doi:10.1016/j.ebiom.2018.09.005. PMID: 30224312.
4.
Anaya YA, Barragan M, Bracho RP, Shaham SH, Bandyopadhyay D, George E, Nguyen D, Tripathi MK. (2025), Proteolysis-targeting chimeras in cancer therapy: Targeted protein degradation for next-generation treatment, Cancer, 131(21):e70132. doi:10.1002/cncr.70132. PMID: 41108678.
5.
Antermite D, Friis SD, Johansson JR, Putra OD, Ackermann L, Johansson MJ. (2023), Late-stage synthesis of heterobifunctional molecules for PROTAC applications via ruthenium-catalysed C‒H amidation, Nat Commun, 14(1):8222. doi:10.1038/s41467-023-43789-9. PMID: 38086825.
6.
Bai N, Miller SA, Andrianov GV, Yates M, Kirubakaran P, Karanicolas J. (2021), Rationalizing PROTAC-Mediated Ternary Complex Formation Using Rosetta, J Chem Inf Model, 61(3):1368-1382. doi:10.1021/acs.jcim.0c01451. PMID: 33625214.
7.
Bekes M, Langley DR, Crews CM. (2022), PROTAC targeted protein degraders: the past is prologue, Nat Rev Drug Discov, 21(3):181-200. doi:10.1038/s41573-021-00371-6. PMID: 35042991.
8.
Bouvier C, Lawrence R, Cavallo F, Xolalpa W, Jordan A, Hjerpe R, Rodriguez MS. (2024), Breaking Bad Proteins-Discovery Approaches and the Road to Clinic for Degraders, Cells, 13(7) doi:10.3390/cells13070578. PMID: 38607017.
9.
Bricelj A, Steinebach C, Kuchta R, Gutschow M, Sosic I. (2021), E3 Ligase Ligands in Successful PROTACs: An Overview of Syntheses and Linker Attachment Points, Front Chem, 9:707317. doi:10.3389/fchem.2021.707317. PMID: 34291038.
10.
Burke MR, Smith AR, Zheng G. (2022), Overcoming Cancer Drug Resistance Utilizing PROTAC Technology, Front Cell Dev Biol, 10:872729. doi:10.3389/fcell.2022.872729. PMID: 35547806.
11.
Cai J, Chen C, Wang J, Zhang X, Cui Y, Zhu Q, Sun H. (2025), PROTAC: a revolutionary technology propelling small molecule drugs into the next golden age, Front Oncol, 15:1676414. doi:10.3389/fonc.2025.1676414. PMID: 41220927.
12.
Christoforou I, Kalatzis A, Siamidi A, Vlachou M, Pispas S, Pippa N. (2025), The Ubiquitous Use of Polyethylene Glycol in Pharmaceutical Design and Development: Technological Aspects and Future Perspectives, Nanomaterials (Basel), 15(23) doi:10.3390/nano15231762. PMID: 41369439.
13.
Crews CM. (2010), Targeting the undruggable proteome: the small molecules of my dreams, Chem Biol, 17(6):551-5. doi:10.1016/j.chembiol.2010.05.011. PMID: 20609404.
14.
Dale B, Cheng M, Park KS, Kaniskan HU, Xiong Y, Jin J. (2021), Advancing targeted protein degradation for cancer therapy, Nat Rev Cancer, 21(10):638-654. doi:10.1038/s41568-021-00365-x. PMID: 34131295.
15.
Danishuddin, Jamal MS, Song KS, Lee KW, Kim JJ, Park YM. (2023), Revolutionizing Drug Targeting Strategies: Integrating Artificial Intelligence and Structure-Based Methods in PROTAC Development, Pharmaceuticals (Basel), 16(12) doi:10.3390/ph16121649. PMID: 38139776.
16.
Diehl CJ, Ciulli A. (2022), Discovery of small molecule ligands for the von Hippel-Lindau (VHL) E3 ligase and their use as inhibitors and PROTAC degraders, Chem Soc Rev, 51(19):8216-8257. doi:10.1039/d2cs00387b. PMID: 35983982.
17.
Ebadi P, Stratton CM, Olsen SK. (2025), E3 ubiquitin ligases in signaling, disease, and therapeutics, Trends Biochem Sci, 50(11):960-976. doi:10.1016/j.tibs.2025.07.009. PMID: 40940201.
18.
Edmondson SD, Yang B, Fallan C. (2019), Proteolysis targeting chimeras (PROTACs) in 'beyond rule-of-five' chemical space: Recent progress and future challenges, Bioorg Med Chem Lett, 29(13):1555-1564. doi:10.1016/j.bmcl.2019.04.030. PMID: 31047748.
19.
Egbert M, Whitty A, Keseru GM, Vajda S. (2019), Why Some Targets Benefit from beyond Rule of Five Drugs, J Med Chem, 62(22):10005-10025. doi:10.1021/acs.jmedchem.8b01732. PMID: 31188592.
20.
Ermondi G, Garcia-Jimenez D, Caron G. (2021), PROTACs and Building Blocks: The 2D Chemical Space in Very Early Drug Discovery, Molecules, 26(3) doi:10.3390/molecules26030672. PMID: 33525371.
21.
Fan G, Chen S, Zhang Q, Yu N, Shen Z, Liu Z, Guo W, Tang Z, Yang J, Liu M. (2025), Proteolysis-Targeting Chimera (PROTAC): Current Applications and Future Directions, MedComm (2020), 6(10):e70401. doi:10.1002/mco2.70401. PMID: 41049269.
22.
Faryal B, Ul Abideen Z, Irfan M, Ahmed H, Jalilov F, Abduraximova L, Ashraf GA. (2026), Targeted Protein Degradation in Cancer: PROTACs, New Targets, and Clinical Mechanisms, Biomolecules, 16(2) doi:10.3390/biom16020325. PMID: 41750393.
23.
Galla MS, Sharma N, Mishra P, Shankaraiah N. (2024), Recent insights of PROTAC developments in inflammation-mediated and autoimmune targets: a critical review, RSC Med Chem, 15(8):2585-2600. doi:10.1039/d4md00142g. PMID: 39149114.
24.
Gioiello L, Di Martino RMC, Pirali T. (2025), Expanding the Scope of PROTACs: Opportunities and Challenges in Topical Delivery, J Med Chem, 68(22):23676-23689. doi:10.1021/acs.jmedchem.5c01911. PMID: 41255358.
25.
Hakem F, Abdelwaly A, Alshaman R, Alattar A, Alanazi FE, Zaitone SA, Helal MA. (2025), Recent Advances in the Development of Pro-PROTAC for Selective Protein Degradation, Pharmaceutics, 17(9) doi:10.3390/pharmaceutics17091160. PMID: 41012497.
26.
Hamilton EP, Jeselsohn RM, Vahdat LT, Hurvitz SA. (2025), PROteolysis TArgeting Chimera (PROTAC) Estrogen Receptor Degraders for Treatment of Estrogen Receptor-Positive Advanced Breast Cancer, Target Oncol, 20(3):431-444. doi:10.1007/s11523-025-01137-5. PMID: 40327300.
27.
Han B. (2020), A suite of mathematical solutions to describe ternary complex formation and their application to targeted protein degradation by heterobifunctional ligands, J Biol Chem, 295(45):15280-15291. doi:10.1074/jbc.RA120.014715. PMID: 32859748.
28.
Han X, Sun Y. (2023), PROTACs: A novel strategy for cancer drug discovery and development, MedComm (2020), 4(3):e290. doi:10.1002/mco2.290. PMID: 37261210.
29.
He S, Dong G, Sheng C. (2025), Strategies for Precise Modulation of Protein Degradation, Acc Chem Res, 58(8):1236-1248. doi:10.1021/acs.accounts.5c00003. PMID: 40132213.
30.
Hong SH, Nguyen T, Ongkingco JF, Nazzaro A, Arora PS. (2025), From Concepts to Inhibitors: A Blueprint for Targeting Protein-Protein Interactions, Chem Rev, 125(14):6819-6869. doi:10.1021/acs.chemrev.5c00046. PMID: 40553022.
31.
Hornberger KR, Araujo EMV. (2023), Physicochemical Property Determinants of Oral Absorption for PROTAC Protein Degraders, J Med Chem, 66(12):8281-8287. doi:10.1021/acs.jmedchem.3c00740. PMID: 37279490.
32.
Jia S, Li Q, Rui X, Qin W, Zhang W, Dou J, Zhang X. (2025), The ubiquitin-proteasome system in Alzheimer's disease: mechanism of action and current status of treatment, Front Aging Neurosci, 17:1730206. doi:10.3389/fnagi.2025.1730206. PMID: 41415890.
33.
Kamaraj R, Ghosh S, Das S, Sen S, Kumar P, Majumdar M, Dasgupta R, Mukherjee S, Das S, Ghose I, Pavek P, Raja Karuppiah MP, Chuturgoon AA, Anand K. (2024), Targeted Protein Degradation (TPD) for Immunotherapy: Understanding Proteolysis Targeting Chimera-Driven Ubiquitin-Proteasome Interactions, Bioconjug Chem, 35(8):1089-1115. doi:10.1021/acs.bioconjchem.4c00253. PMID: 38990186.
34.
Kandel R, Jung J, Neal S. (2024), Proteotoxic stress and the ubiquitin proteasome system, Semin Cell Dev Biol, 156:107-120. doi:10.1016/j.semcdb.2023.08.002. PMID: 37734998.
35.
Kim G, Grams RJ, Hsu KL. (2025), Advancing Covalent Ligand and Drug Discovery beyond Cysteine, Chem Rev, 125(14):6653-6684. doi:10.1021/acs.chemrev.5c00001. PMID: 40404146.
36.
Kim H, Park J, Kim JM. (2022), Targeted Protein Degradation to Overcome Resistance in Cancer Therapies: PROTAC and N-Degron Pathway, Biomedicines, 10(9) doi:10.3390/biomedicines10092100. PMID: 36140200.
37.
Koirala M, Yan L, Mohamed Z, DiPaola M. (2025), AI-Integrated QSAR Modeling for Enhanced Drug Discovery: From Classical Approaches to Deep Learning and Structural Insight, Int J Mol Sci, 26(19) doi:10.3390/ijms26199384. PMID: 41096653.
38.
Konstantinidou M, Li J, Zhang B, Wang Z, Shaabani S, Ter Brake F, Essa K, Domling A. (2019), PROTACs- a game-changing technology, Expert Opin Drug Discov, 14(12):1255-1268. doi:10.1080/17460441.2019.1659242. PMID: 31538491.
39.
Kubryn N, Fijalkowski L, Nowaczyk J, Jamil A, Nowaczyk A. (2025), PROTAC Technology as a New Tool for Modern Pharmacotherapy, Molecules, 30(10) doi:10.3390/molecules30102123. PMID: 40430296.
40.
Kudo G, Hirao T, Harada R, Shigeta Y, Hirokawa T, Yoshino R. (2025), Construction of PROTAC-Mediated Ternary Complex Structure Distribution Profiles Using Extensive Conformational Search, J Chem Inf Model, 65(13):6939-6948. doi:10.1021/acs.jcim.5c00102. PMID: 40550492.
41.
Lai AC, Crews CM. (2017), Induced protein degradation: an emerging drug discovery paradigm, Nat Rev Drug Discov, 16(2):101-114. doi:10.1038/nrd.2016.211. PMID: 27885283.
42.
Lee J, Lee Y, Jung YM, Park JH, Yoo HS, Park J. (2022), Discovery of E3 Ligase Ligands for Target Protein Degradation, Molecules, 27(19) doi:10.3390/molecules27196515. PMID: 36235052.
43.
Li F, Hu Q, Zhang X, Sun R, Liu Z, Wu S, Tian S, Ma X, Dai Z, Yang X, Gao S, Bai F. (2022a), DeepPROTACs is a deep learning-based targeted degradation predictor for PROTACs, Nat Commun, 13(1):7133. doi:10.1038/s41467-022-34807-3. PMID: 36414666.
44.
Li K, Crews CM. (2022), PROTACs: past, present and future, Chem Soc Rev, 51(12):5214-5236. doi:10.1039/d2cs00193d. PMID: 35671157.
45.
Li X, Pu W, Zheng Q, Ai M, Chen S, Peng Y. (2022b), Proteolysis-targeting chimeras (PROTACs) in cancer therapy, Mol Cancer, 21(1):99. doi:10.1186/s12943-021-01434-3. PMID: 35410300.
46.
Lin CT, Shiau YP, Lin CC. (2026), Machine learning in targeted protein degradation drug design: a technical review of PROTACs and molecular glues, Drug Discov Today, 31(1):104563. doi:10.1016/j.drudis.2025.104563. PMID: 41318024.
47.
Liu J, Roy MJ, Isbel L, Li F. (2025), Accurate PROTAC-targeted degradation prediction with DegradeMaster, Bioinformatics, 41(Supplement_1):i342-i351. doi:10.1093/bioinformatics/btaf191. PMID: 40662822.
48.
Liu Y, Liang J, Zhu R, Yang Y, Wang Y, Wei W, Li H, Chen L. (2024), Application of PROTACs in Target Identification and Target Validation, Acta Mater Med, 3(1):72-87. doi:10.15212/amm-2024-0010. PMID: 39373008.
49.
Liu Y, Yang J, Wang T, Luo M, Chen Y, Chen C, Ronai Z, Zhou Y, Ruppin E, Han L. (2023), Expanding PROTACtable genome universe of E3 ligases, Nat Commun, 14(1):6509. doi:10.1038/s41467-023-42233-2. PMID: 37845222.
50.
Liu Y, Zhang X, Chen X, Zhang F. (2026), Proteolysis-targeting chimera (PROTAC) nanomedicines toward cancer treatment: From synthesis to therapeutic delivery, Biomaterials, 325:123621. doi:10.1016/j.biomaterials.2025.123621. PMID: 40819599.
51.
Liu Z, Hu M, Yang Y, Du C, Zhou H, Liu C, Chen Y, Fan L, Ma H, Gong Y, Xie Y. (2022), An overview of PROTACs: a promising drug discovery paradigm, Mol Biomed, 3(1):46. doi:10.1186/s43556-022-00112-0. PMID: 36536188.
52.
Luo H, Tian Y, Abdullah R, Zhang B, Ma Y, Zhang G. (2025), Advancing Design Strategy of PROTACs for Cancer Therapy, MedComm (2020), 6(7):e70258. doi:10.1002/mco2.70258. PMID: 40567248.
53.
Ma Z, Zhou J. (2025), NDA Submission of Vepdegestrant (ARV-471) to U.S. FDA: The Beginning of a New Era of PROTAC Degraders, J Med Chem, 68(14):14129-14136. doi:10.1021/acs.jmedchem.5c01818. PMID: 40702893.
54.
Mancarella C, Morrione A, Scotlandi K. (2023), PROTAC-Based Protein Degradation as a Promising Strategy for Targeted Therapy in Sarcomas, Int J Mol Sci, 24(22) doi:10.3390/ijms242216346. PMID: 38003535.
55.
Martin-Acosta P, Xiao X. (2021), PROTACs to address the challenges facing small molecule inhibitors, Eur J Med Chem, 210:112993. doi:10.1016/j.ejmech.2020.112993. PMID: 33189436.
56.
Melvin AT, Woss GS, Park JH, Waters ML, Allbritton NL. (2013), Measuring activity in the ubiquitin-proteasome system: from large scale discoveries to single cells analysis, Cell Biochem Biophys, 67(1):75-89. doi:10.1007/s12013-013-9621-9. PMID: 23686610.
57.
Michaelides IN, Collie GW. (2023), E3 Ligases Meet Their Match: Fragment-Based Approaches to Discover New E3 Ligands and to Unravel E3 Biology, J Med Chem, 66(5):3173-3194. doi:10.1021/acs.jmedchem.2c01882. PMID: 36821822.
58.
Mohapatra P, Gopikrishnan M, Doss CG, Chandrasekaran N. (2024), How Precise are Nanomedicines in Overcoming the Blood-Brain Barrier? A Comprehensive Review of the Literature, Int J Nanomedicine, 19:2441-2467. doi:10.2147/IJN.S442520. PMID: 38482521.
59.
Moon Y, Jeon SI, Shim MK, Kim K. (2023), Cancer-Specific Delivery of Proteolysis-Targeting Chimeras (PROTACs) and Their Application to Cancer Immunotherapy, Pharmaceutics, 15(2) doi:10.3390/pharmaceutics15020411. PMID: 36839734.
60.
Mostofian B, Martin HJ, Razavi A, Patel S, Allen B, Sherman W, Izaguirre JA. (2023), Targeted Protein Degradation: Advances, Challenges, and Prospects for Computational Methods, J Chem Inf Model, 63(17):5408-5432. doi:10.1021/acs.jcim.3c00603. PMID: 37602861.
61.
Nalawansha DA, Crews CM. (2020), PROTACs: An Emerging Therapeutic Modality in Precision Medicine, Cell Chem Biol, 27(8):998-1014. doi:10.1016/j.chembiol.2020.07.020. PMID: 32795419.
62.
Nunes J, McGonagle GA, Eden J, Kiritharan G, Touzet M, Lewell X, Emery J, Eidam H, Harling JD, Anderson NA. (2019), Targeting IRAK4 for Degradation with PROTACs, ACS Med Chem Lett, 10(7):1081-1085. doi:10.1021/acsmedchemlett.9b00219. PMID: 31312412.
63.
Osman J, Thompson PE, Jorg M, Scanlon MJ. (2025), Methods to accelerate PROTAC drug discovery, Biochem J, 482(13):921-37. doi:10.1042/BCJ20243018. PMID: 40570202.
64.
Paiva SL, Crews CM. (2019), Targeted protein degradation: elements of PROTAC design, Curr Opin Chem Biol, 50:111-119. doi:10.1016/j.cbpa.2019.02.022. PMID: 31004963.
65.
Park KS, Jeon M. (2025), Advancing PROTAC Discovery Through Artificial Intelligence: Opportunities, Challenges, and Future Directions, Pharmaceuticals (Basel), 18(12) doi:10.3390/ph18121793. PMID: 41471282.
66.
Pettersson M, Crews CM. (2019), PROteolysis TArgeting Chimeras (PROTACs) - Past, present and future, Drug Discov Today Technol, 31:15-27. doi:10.1016/j.ddtec.2019.01.002. PMID: 31200855.
67.
Qi SM, Dong J, Xu ZY, Cheng XD, Zhang WD, Qin JJ. (2021), PROTAC: An Effective Targeted Protein Degradation Strategy for Cancer Therapy, Front Pharmacol, 12:692574. doi:10.3389/fphar.2021.692574. PMID: 34025443.
68.
Rej RK, Allu SR, Roy J, Acharyya RK, Kiran INC, Addepalli Y, Dhamodharan V. (2024), Orally Bioavailable Proteolysis-Targeting Chimeras: An Innovative Approach in the Golden Era of Discovering Small-Molecule Cancer Drugs, Pharmaceuticals (Basel), 17(4) doi:10.3390/ph17040494. PMID: 38675453.
69.
Rui H, Ashton KS, Min J, Wang C, Potts PR. (2023), Protein-protein interfaces in molecular glue-induced ternary complexes: classification, characterization, and prediction, RSC Chem Biol, 4(3):192-215. doi:10.1039/d2cb00207h. PMID: 36908699.
70.
Rutherford KA, McManus KJ. (2024), PROTACs: Current and Future Potential as a Precision Medicine Strategy to Combat Cancer, Mol Cancer Ther, 23(4):454-463. doi:10.1158/1535-7163.MCT-23-0747. PMID: 38205881.
71.
Scott DC, Dharuman S, Griffith E, Chai SC, Ronnebaum J, King MT, Tangallapally R, Lee C, Gee CT, Yang L, Li Y, Loudon VC, Lee HW, Ochoada J, Miller DJ, Jayasinghe T, Paulo JA, Elledge SJ, Harper JW, Chen T, Lee RE, Schulman BA. (2024), Principles of paralog-specific targeted protein degradation engaging the C-degron E3 KLHDC2, Nat Commun, 15(1):8829. doi:10.1038/s41467-024-52966-3. PMID: 39396041.
72.
Sincere NI, Anand K, Ashique S, Yang J, You C. (2023), PROTACs: Emerging Targeted Protein Degradation Approaches for Advanced Druggable Strategies, Molecules, 28(10) doi:10.3390/molecules28104014. PMID: 37241755.
73.
Snyder LB, Neklesa TK, Willard RR, Gordon DA, Pizzano J, Vitale N, Robling K, Dorso MA, Moghrabi W, Landrette S, Gedrich R, Lee SH, Taylor ICA, Houston JG. (2025), Preclinical Evaluation of Bavdegalutamide (ARV-110), a Novel PROteolysis TArgeting Chimera Androgen Receptor Degrader, Mol Cancer Ther, 24(4):511-522. doi:10.1158/1535-7163.MCT-23-0655. PMID: 39670468.
74.
Stewart MD, Ritterhoff T, Klevit RE, Brzovic PS. (2016), E2 enzymes: more than just middle men, Cell Res, 26(4):423-40. doi:10.1038/cr.2016.35. PMID: 27002219.
75.
Syahputra EW, Lee H, Cho H, Park HJ, Park KS, Hwang D. (2025), PROTAC Delivery Strategies for Overcoming Physicochemical Properties and Physiological Barriers in Targeted Protein Degradation, Pharmaceutics, 17(4) doi:10.3390/pharmaceutics17040501. PMID: 40284496.
76.
Troup RI, Fallan C, Baud MGJ. (2020), Current strategies for the design of PROTAC linkers: a critical review, Explor Target Antitumor Ther, 1(5):273-312. doi:10.37349/etat.2020.00018. PMID: 36046485.
77.
Tunjic TM, Weber N, Brunsteiner M. (2023), Computer aided drug design in the development of proteolysis targeting chimeras, Comput Struct Biotechnol J, 21:2058-2067. doi:10.1016/j.csbj.2023.02.042. PMID: 36968015.
78.
Wang C, Zhang Y, Chen W, Wu Y, Xing D. (2024a), New-generation advanced PROTACs as potential therapeutic agents in cancer therapy, Mol Cancer, 23(1):110. doi:10.1186/s12943-024-02024-9. PMID: 38773495.
79.
Wang H, Peng J, Li H, Lan Y, Guo J, Qiu Q, Huang X. (2025a), E3 Ubiquitin Ligases: Structures, Biological Functions, Diseases, and Therapy, MedComm (2020), 6(12):e70528. doi:10.1002/mco2.70528. PMID: 41362701.
80.
Wang Q, Sun B, Yi Y, Velkov T, Shen J, Dai C, Jiang H. (2025b), Progress of AI-Driven Drug-Target Interaction Prediction and Lead Optimization, Int J Mol Sci, 26(20) doi:10.3390/ijms262010037. PMID: 41155330.
81.
Wang S, He F, Tian C, Sun A. (2024b), From PROTAC to TPD: Advances and Opportunities in Targeted Protein Degradation, Pharmaceuticals (Basel), 17(1) doi:10.3390/ph17010100. PMID: 38256933.
82.
Wang Y, Jiang X, Feng F, Liu W, Sun H. (2020), Degradation of proteins by PROTACs and other strategies, Acta Pharm Sin B, 10(2):207-238. doi:10.1016/j.apsb.2019.08.001. PMID: 32082969.
83.
Wang Z, Zhang D, Inuzuka H, Wei W. (2025c), PROTAC technology for prostate cancer treatment, Acta Mater Med, 4(1):99-121. doi:10.15212/amm-2024-0075. PMID: 40832004.
84.
Wu X, Shu Y, Zheng Y, Zhang P, Cong H, Zou Y, Cai H, Zha Z. (2025), Recent Advances in Nanomedicine: Cutting-Edge Research on Nano-PROTAC Delivery Systems for Cancer Therapy, Pharmaceutics, 17(8) doi:10.3390/pharmaceutics17081037. PMID: 40871058.
85.
Xi JY, Zhang RY, Chen K, Yao L, Li MQ, Jiang R, Li XY, Fan L. (2022), Advances and perspectives of proteolysis targeting chimeras (PROTACs) in drug discovery, Bioorg Chem, 125:105848. doi:10.1016/j.bioorg.2022.105848. PMID: 35533582.
86.
Xie X, Yu T, Li X, Zhang N, Foster LJ, Peng C, Huang W, He G. (2023), Recent advances in targeting the "undruggable" proteins: from drug discovery to clinical trials, Signal Transduct Target Ther, 8(1):335. doi:10.1038/s41392-023-01589-z. PMID: 37669923.
87.
Yao D, Li T, Yu L, Hu M, He Y, Zhang R, Wu J, Li S, Kuang W, Yang X, Liu G, Xie Y. (2024), Selective degradation of hyperphosphorylated tau by proteolysis-targeting chimeras ameliorates cognitive function in Alzheimer's disease model mice, Front Pharmacol, 15:1351792. doi:10.3389/fphar.2024.1351792. PMID: 38919259.
88.
Yao T, Xiao H, Wang H, Xu X. (2022), Recent Advances in PROTACs for Drug Targeted Protein Research, Int J Mol Sci, 23(18) doi:10.3390/ijms231810328. PMID: 36142231.
89.
Yim J, Park J, Kim G, Lee HH, Chung JS, Jo A, Koh M, Park J. (2024), Conditional PROTAC: Recent Strategies for Modulating Targeted Protein Degradation, ChemMedChem, 19(22):e202400326. doi:10.1002/cmdc.202400326. PMID: 38993102.
90.
Zhang G, Yan S, Liu Y, Du Z, Min Q, Qin S. (2025), PROTACs coupled with oligonucleotides to tackle the undruggable, Bioanalysis, 17(4):261-276. doi:10.1080/17576180.2025.2459528. PMID: 39895280.
91.
Zhao C, Dekker FJ. (2022), Novel Design Strategies to Enhance the Efficiency of Proteolysis Targeting Chimeras, ACS Pharmacol Transl Sci, 5(9):710-723. doi:10.1021/acsptsci.2c00089. PMID: 36110375.
92.
Zhao L, Zhao J, Zhong K, Tong A, Jia D. (2022), Targeted protein degradation: mechanisms, strategies and application, Signal Transduct Target Ther, 7(1):113. doi:10.1038/s41392-022-00966-4. PMID: 35379777.
93.
Zhao M, Li B, Gao Y, Zhang R, Ahmattohti S, Li J, Shi X. (2026), Linker Engineering in Stapled Peptides for Enhanced Membrane Permeability: Screening and Optimization Strategies, Int J Mol Sci, 27(7) doi:10.3390/ijms27073077. PMID: 41977264.
94.
Zhong G, Chang X, Xie W, Zhou X. (2024), Targeted protein degradation: advances in drug discovery and clinical practice, Signal Transduct Target Ther, 9(1):308. doi:10.1038/s41392-024-02004-x. PMID: 39500878.
95.
Zhou Q, Wang W, Deng C. (2025), Advancements in Proteolysis Targeting Chimeras for Targeted Therapeutic Strategies in Alzheimer's Disease, Mol Neurobiol, 62(8):9686-9709. doi:10.1007/s12035-025-04838-0. PMID: 40133753.
96.
Zou Y, Ma D, Wang Y. (2019), The PROTAC technology in drug development, Cell Biochem Funct, 37(1):21-30. doi:10.1002/cbf.3369. PMID: 30604499.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.