About the Journal
Scope of the Journal
Indexed In
Journal Information
Current issue
Archive
Editorial Board
Journal Policy
APC Policy
Authorship Policy
Author Recognition Policy
Citation Policy
Conflict of Interest (COI) Policy
Copyright Policy
Data Availability Policy
DEI Policy
Digital Archiving Policy
Ethical Approval Policy
Funding Policy
Image Policy
Open Access Policy
Ownership & Governance
Peer Review Policy
Personal Data Protection Policy
Plagiarism Policy
Post-Publication Policy
Preprint Policy
Author Instructions
Submit Manuscript
Contact Information
Journal Policy
APC Policy
Authorship Policy
Author Recognition Policy
Citation Policy
Conflict of Interest (COI) Policy
Copyright Policy
Data Availability Policy
DEI Policy
Digital Archiving Policy
Ethical Approval Policy
Funding Policy
Image Policy
Open Access Policy
Ownership & Governance
Peer Review Policy
Personal Data Protection Policy
Plagiarism Policy
Post-Publication Policy
Preprint Policy
REVIEW PAPER
MODELLER-Based Homology Modeling: A Critical Review of Protocols, Advances, and Computational Challenges
1
Department of Biotechnology, Lady Doak College, Madurai, India
2
Department of Zoology and Research Centre, Lady Doak College, Madurai, India
Submission date: 2025-08-30
Final revision date: 2025-09-24
Acceptance date: 2025-09-24
Online publication date: 2026-01-28
Publication date: 2026-01-28
Corresponding author
Thangamalai Mowna Sundari
Department of Zoology and Research Centre, Lady Doak College, Madurai, 625002, Madurai, India
Department of Zoology and Research Centre, Lady Doak College, Madurai, 625002, Madurai, India
Journal of Medico Informatics 2026;02(Issue 01):40-47
HIGHLIGHTS
- MODELLER remains essential for template-based protein structure prediction and comparative modeling.
- Multi-template modeling improves structural accuracy in low sequence identity proteins.
- DOPE scoring enhances model validation and structural reliability assessment.
- Hybrid approaches combining MODELLER and AlphaFold improve prediction accuracy.
- Challenges remain in low identity modeling, flexibility prediction, and computational efficiency.
KEYWORDS
TOPICS
ABSTRACT
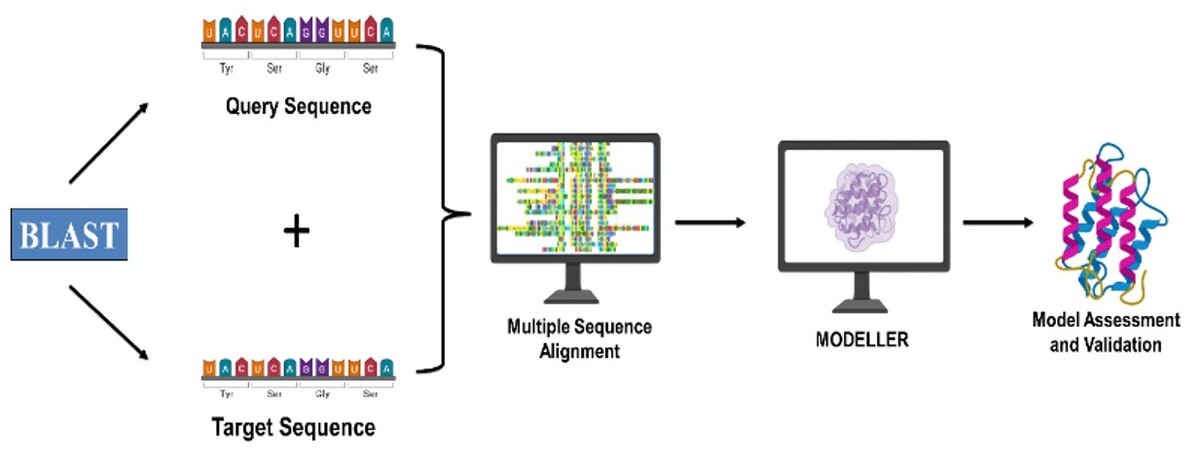
Homology modeling using MODELLER has been a cornerstone of computational structural biology for over three decades, yet its protocols, advances, and computational challenges have not been fully reviewed in the context of modern structural prediction methods. Despite breakthroughs from deep learning approaches like AlphaFold, MODELLER’s satisfaction of spatial restraints methodology remains vital in template-based modeling, especially in low sequence identity cases, multi-template strategies, and specialized applications.
This review critically examines current MODELLER protocols, recent methodological improvements, and persistent computational challenges. We analyzed literature on MODELLER applications, benchmarking, and algorithmic refinements, focusing on template selection, alignment optimization, model validation, and integration with modern computational methods.
Our analysis shows that although MODELLER’s core algorithm has remained largely unchanged since the 1990s, advances in template selection, multi-template modeling, and statistical potentials such as DOPE scoring have improved model accuracy. Recent work highlights the impact of optimized σ value estimation and incorporation of statistical potential terms, particularly in multi-template modeling. Nonetheless, challenges persist, including reduced accuracy in low sequence identity modeling, scalability issues, and poor handling of conformational flexibility.
MODELLER retains unique strengths in template-based modeling, particularly when combined with modern validation strategies and machine learning–assisted template selection. Looking ahead, progress in hybrid modeling approaches, more accurate scoring functions, and greater computational efficiency will be essential for MODELLER to sustain its relevance within the rapidly evolving field of protein structure prediction.
ACKNOWLEDGEMENTS
The authors thankfully acknowledge the Lady Doak College, Madurai, Tamil Nadu, India for providing necessary facilities for performing this review work.
FUNDING
No funding is reported with this review work.
CONFLICT OF INTEREST
The authors declare that they have no known financial, personal, academic, or other relationships that could inappropriately influence, or be perceived to influence, the work reported in this manuscript. All authors confirm that there are no competing interests to declare.
PEER REVIEW INFORMATION
Article has been screened for originality
REFERENCES (30)
1.
Bender BJ, Marlow B, Meiler J. (2020), Improving homology modeling from low-sequence identity templates in Rosetta: A case study in GPCRs, PLoS Comput Biol, 16(10):e1007597. doi:10.1371/journal.pcbi.1007597. PMID: 33112852.
2.
Binbay FA, Rathod DC, George AAP, Imhof D. (2023), Quality Assessment of Selected Protein Structures Derived from Homology Modeling and AlphaFold, Pharmaceuticals (Basel), 16(12) doi:10.3390/ph16121662. PMID: 38139789.
3.
Chen L, Li Q, Nasif KFA, Xie Y, Deng B, Niu S, Pouriyeh S, Dai Z, Chen J, Xie CY. (2024), AI-Driven Deep Learning Techniques in Protein Structure Prediction, Int J Mol Sci, 25(15) doi:10.3390/ijms25158426. PMID: 39125995.
4.
Cieslik M, Mura C. (2011), A lightweight, flow-based toolkit for parallel and distributed bioinformatics pipelines, BMC Bioinformatics, 12:61. doi:10.1186/1471-2105-12-61. PMID: 21352538.
5.
Eramian D, Shen MY, Devos D, Melo F, Sali A, Marti-Renom MA. (2006), A composite score for predicting errors in protein structure models, Protein Sci, 15(7):1653-66. doi:10.1110/ps.062095806. PMID: 16751606.
6.
Eswar N, Webb B, Marti-Renom MA, Madhusudhan MS, Eramian D, Shen MY, Pieper U, Sali A. (2006), Comparative protein structure modeling using Modeller, Curr Protoc Bioinformatics, Chapter 5:Unit-5 6. doi:10.1002/0471250953.bi0506s15. PMID: 18428767.
7.
Fiser A. (2010), Template-based protein structure modeling, Methods Mol Biol, 673:73-94. doi:10.1007/978-1-60761-842-3_6. PMID: 20835794.
8.
Gil Zuluaga FH, D'Arminio N, Bardozzo F, Tagliaferri R, Marabotti A. (2023), An automated pipeline integrating AlphaFold 2 and MODELLER for protein structure prediction, Comput Struct Biotechnol J, 21:5620-5629. doi:10.1016/j.csbj.2023.10.056. PMID: 38047234.
9.
Grosdidier S, Totrov M, Fernandez-Recio J. (2009), Computer applications for prediction of protein-protein interactions and rational drug design, Adv Appl Bioinform Chem, 2:101-23. PMID: 21918619.
10.
Guedes IA, Pereira FSS, Dardenne LE. (2018), Empirical Scoring Functions for Structure-Based Virtual Screening: Applications, Critical Aspects, and Challenges, Front Pharmacol, 9:1089. doi:10.3389/fphar.2018.01089. PMID: 30319422.
11.
Haddad Y, Adam V, Heger Z. (2020), Ten quick tips for homology modeling of high-resolution protein 3D structures, PLoS Comput Biol, 16(4):e1007449. doi:10.1371/journal.pcbi.1007449. PMID: 32240155.
12.
Heo L, Feig M. (2024), One bead per residue can describe all-atom protein structures, Structure, 32(1):97-111 e6. doi:10.1016/j.str.2023.10.013. PMID: 38000367.
13.
Janson G, Grottesi A, Pietrosanto M, Ausiello G, Guarguaglini G, Paiardini A. (2019), Revisiting the "satisfaction of spatial restraints" approach of MODELLER for protein homology modeling, PLoS Comput Biol, 15(12):e1007219. doi:10.1371/journal.pcbi.1007219. PMID: 31846452.
14.
Joo K, Lee J, Seo JH, Lee K, Kim BG, Lee J. (2009), All-atom chain-building by optimizing MODELLER energy function using conformational space annealing, Proteins, 75(4):1010-23. doi:10.1002/prot.22312. PMID: 19089941.
15.
Koehler Leman J, Ulmschneider MB, Gray JJ. (2015), Computational modeling of membrane proteins, Proteins, 83(1):1-24. doi:10.1002/prot.24703. PMID: 25355688.
16.
Kryshtafovych A, Schwede T, Topf M, Fidelis K, Moult J. (2019), Critical assessment of methods of protein structure prediction (CASP)-Round XIII, Proteins, 87(12):1011-1020. doi:10.1002/prot.25823. PMID: 31589781.
17.
Larsson P, Wallner B, Lindahl E, Elofsson A. (2008), Using multiple templates to improve quality of homology models in automated homology modeling, Protein Sci, 17(6):990-1002. doi:10.1110/ps.073344908. PMID: 18441233.
18.
Meng EC, Goddard TD, Pettersen EF, Couch GS, Pearson ZJ, Morris JH, Ferrin TE. (2023), UCSF ChimeraX: Tools for structure building and analysis, Protein Sci, 32(11):e4792. doi:10.1002/pro.4792. PMID: 37774136.
19.
Miller HE, Montemayor D, Abdul J, Vines A, Levy SA, Hartono SR, Sharma K, Frost B, Chedin F, Bishop AJR. (2022), Quality-controlled R-loop meta-analysis reveals the characteristics of R-loop consensus regions, Nucleic Acids Res, 50(13):7260-7286. doi:10.1093/nar/gkac537. PMID: 35758606.
20.
Pearce R, Zhang Y. (2021), Toward the solution of the protein structure prediction problem, J Biol Chem, 297(1):100870. doi:10.1016/j.jbc.2021.100870. PMID: 34119522.
21.
Sali A, Blundell TL. (1993), Comparative protein modelling by satisfaction of spatial restraints, J Mol Biol, 234(3):779-815. doi:10.1006/jmbi.1993.1626. PMID: 8254673.
22.
Seffernick JT, Lindert S. (2020), Hybrid methods for combined experimental and computational determination of protein structure, J Chem Phys, 153(24):240901. doi:10.1063/5.0026025. PMID: 33380110.
23.
Shen MY, Sali A. (2006), Statistical potential for assessment and prediction of protein structures, Protein Sci, 15(11):2507-24. doi:10.1110/ps.062416606. PMID: 17075131.
24.
Szilagyi A, Zhang Y. (2014), Template-based structure modeling of protein-protein interactions, Curr Opin Struct Biol, 24:10-23. doi:10.1016/j.sbi.2013.11.005. PMID: 24721449.
25.
Varadi M, Tsenkov M, Velankar S. (2025), Challenges in bridging the gap between protein structure prediction and functional interpretation, Proteins, 93(1):400-410. doi:10.1002/prot.26614. PMID: 37850517.
26.
Vyas VK, Ukawala RD, Ghate M, Chintha C. (2012), Homology modeling a fast tool for drug discovery: current perspectives, Indian J Pharm Sci, 74(1):1-17. doi:10.4103/0250-474X.102537. PMID: 23204616.
27.
Wang T, Jin X, Lu X, Min X, Ge S, Li S. (2023), Empirical validation of ProteinMPNN's efficiency in enhancing protein fitness, Front Genet, 14:1347667. doi:10.3389/fgene.2023.1347667. PMID: 38274106.
28.
Webb B, Sali A. (2016), Comparative Protein Structure Modeling Using MODELLER, Curr Protoc Bioinformatics, 54:5 6 1-5 6 37. doi:10.1002/cpbi.3. PMID: 27322406.
29.
Wlodawer A. (2017), Stereochemistry and Validation of Macromolecular Structures, Methods Mol Biol, 1607:595-610. doi:10.1007/978-1-4939-7000-1_24. PMID: 28573590.
30.
Xu X, Duan R, Zou X. (2023), Template-guided method for protein-ligand complex structure prediction: Application to CASP15 protein-ligand studies, Proteins, 91(12):1829-1836. doi:10.1002/prot.26535. PMID: 37283068.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.